
While we use soap every day, few people know the difference between liquid and bar soap. We have the answers and will dive into ingredients, effectiveness, pH values, glycerin, toxins, and environmental aspects.
Bar soap is made with sodium hydroxide (lye) and liquid soap is made with potassium hydroxide (potash). Liquid soap likely has a better pH whereas bar soap contains more glycerin & superfat. While they are equally effective, bar soap contains fewer chemicals and is more environmentally friendly.
Let’s start with the ingredients.
1. Ingredient Check!
The difference between liquid and bar soap lies in their ingredients. Ordinary soap is made by combining fats or oils and an alkali, such as lye. Bar soap is made with sodium hydroxide (lye) and liquid soap is made with potassium hydroxide (another type of lye, also called potash or KOH). [1], [2]
Next to soap, we have detergents. And while we commonly call a hand wash liquid “soap“, it might actually be detergent! According to the FDA, today, there are very few true soaps on the market. [3]
Synthetic detergents are made in chemical plants and also called surfactants, which is short for “SURFace ACTive AgeNt”. [4] Detergent cleansers are popular because they make suds easily in water and don’t form gummy deposits. Some of these detergent products are actually marketed as “soap” but are not true soap according to the FDA’s regulatory definition. [5]
Finally, there are “soap free soaps” or syndet bars. You also find them labeled as “facial bars”. The word syndet comes from SYNthetic DETergent. Syndet bars are quite simply detergents with the look of bar soap. They have a pH level close to the ideal (slightly acid) and are often recommended by dermatologists. [6], [7], [8]
2. They Are Equally Effective
Liquid soap and bar soap are equally effective. According to studies, handwashing with water alone reduces the presence of bacteria to 23%. If we use plain soap and water, the presence of bacteria is further reduced to 8%. [9]
Despite the rumors, bar soap does not transfer bacteria from person to person and bacteria cannot grow on a bar of soap. [10], [11]

>> Read more: Can shampoo really noursih hair?
Is bar soap and liquid soap antibacterial?
Antibacterial soaps, also called antimicrobial or antiseptic soaps, contain certain chemicals not found in natural soaps. It’s the antibacterial agent triclosan, that is most commonly added to reduce or prevent bacterial infection.
However, studies show, that antibacterial soap is no more effective in reducing bacteria than regular soap. [12], [13], [14], [15], [16]
“I suspect there are a lot of consumers who assume that by using an antibacterial soap product, they are protecting themselves from illness, protecting their families. But we don’t have any evidence that that is really the case over simple soap and water.” [17]
Sandra Kweder, deputy director of the FDA’s drug center
3. Bar Soap Lasts Longer
Researchers found that when washing, we use more liquid soap than bar soap. Specifically, we use more than 6x as much liquid soap than bar soap within a one-time hand washing. That means bar soap will last us much longer. [18]
4. Liquid Soap May Have The Better pH
Studies show, that the skin’s pH is one of the most important biophysical parameters for healthy skin. [19] The pH is a scale used to specify how acidic or basic something is. Anything between 0 and 6.9 is acidic, 7 is neutral, and anything between 7.1 and 14 is considered alkaline. Studies show, that normal skin surface pH is between 4 and 5 in healthy people. [20], [21]
Therefore, the ideal pH of a liquid or bar soap should be acidic, that means it should have a pH of ≤ 5. [22]
Our skin is the first line of defense against microorganisms, wind, pollutants and more. It is our skins acid mantle that really does the job. The acid mantle is a fine film with a slightly acidic pH on the surface of the skin and provides ideal protection.
The soaps commonly used by the population at large have a pH outside the range of normal skin pH values. The majority of soaps have a pH within the range of 9-10. [23]
If we use alkaline products, the skin’s acid mantle will be damaged, the protective acidity is lost, and the skin becomes more prone to damage and infection. Once damaged, it can take up to 14 hours to restore. This begins to erode your skin’s structure. It runs dry and looses its hydration.

>> Learn more about the pH of shampoo
Why are alkaline products so bad?
Because this is the foundation of many skin disorders, like acne, psoriasis and eczema. Studies show skin disorders like acne occur more often when our skin pH is more on the alkaline side. Which is the case when we use highly alkaline products. [24]
Remember: The closer a products pH is to our skin, the healthier it is! [25]
Generally, you will find more liquid soaps and syndet bars with an acidic pH on the market than regular bar soaps.
>> See our recommended liquid soaps.
5. Bar Soap Contains Glycerin & Superfat
During the soap making process (saponification), glycerin is made as a natural by-product. By default, soaps have glycerin in them, about 5-10%. Glycerin pulls water into the outer layer of your skin and traps it there. That means, it effectively hydrates and moisturizes our skin. [26]
As you can imagine, glycerin is a valuable component in soap – and when extracted, the soap no longer contains hydrating and moisturizing features. But exactly this is done. Soap manufacturers extract the glycerin from the soap and put it in lotions and creams. The resulting drying effect of the soap has us come back for lotion to fix the problem.
“Soap isn’t necessarily bad for acne, but the wrong type of soap or cleanser can irritate the skin, making acne worse or making it difficult to use appropriate acne medications — many of which are drying.” [27]
Dr. Kathleen Welsh
Look for natural soaps, that still contain their glycerin as they won’t dry your skin but hydrate and moisturize it. [28]
Natural soaps also use skin beneficial herbs, oils, butters, and aromas which provide even more moisture than glycerin alone. Moreover, many natural soap producers use a process called superfatting in which they either add an extra amount of oil or reduce the amount of lye resulting in about 1-20% extra nourishing “superfat”.

>> Find our moisturizing Dead Sea mud soaps her.
6. Liquid Soaps Go Heavy On Toxins
Depending on the brand, conventional bar and liquid soaps contain loads of chemicals. Many US companies use ingredients that are banned in other countries for their toxicity. Let’s see what they are. Overall, you are more likely to find toxins in liquid soaps than bar soaps.
1. Antibacterial Agents
Because of the way the antibacterial agent triclosan reacts in cells, it causes some bacteria to mutate and become resistant to commonly used drugs. [29] The antibacterial agent triclosan in antibacterial hand soap acts as a hormone disruptor, can increase your waistline, causes the emergence of antibiotic resistant bacteria, and can reduce antibiotic efficacy by 100 times. [30], [31], [32], [33], [34]
The Bad Boys: Triclosan.
The Take-Away: Use natural soap with minimal ingredients and stay away from antibacterial products.
2. Fragrances
When you see “fragrance” or “parfum” on the ingredients list, they really mean phthaltes. Phthaltes disrupt our hormones and can harm our reproductive system. In children, they can disturb normal development. [35], [36], [37]
The Bad Boys: Phthaltes (labeled as parfum or fragrance).
The Take-Away: Go fragrance free or choose products with essential oils.
3. Surfactants
Surfactants increase the spreading properties and some produce bubbles. During production 1,4-dioxane is formed which is known to cause cancer. It is banned from use in the European Union. Short-term exposure may cause eye, nose, and throat irritation, long-term exposure may cause kidney and liver damage. [38], [39]
The Bad Boys: Sodium lauryl sulfate (SLS), sodium laureth sulfate (SLES), cocamidopropyl betaine, and cocamide DEA.
The Take-Away: Go sulfate-free! Non-toxic, safe surfactants include decyl glucoside and laurel glucoside.

4. Preservatives
To extend shelf life, many products contain preservatives. Some may cause breast cancer, are associated with immunotoxicity and allergies, or are neurotoxins. [40], [41], [42]
The Bad Boys: Parabens, benzyl alcohol, methylisothiazolinone, and methylchloroisothiazonlinone.
The Take-Away: Choosing natural bar soap is a simple way to avoid preservatives.
>> Check out our RESOURCES for safe, non-toxic products
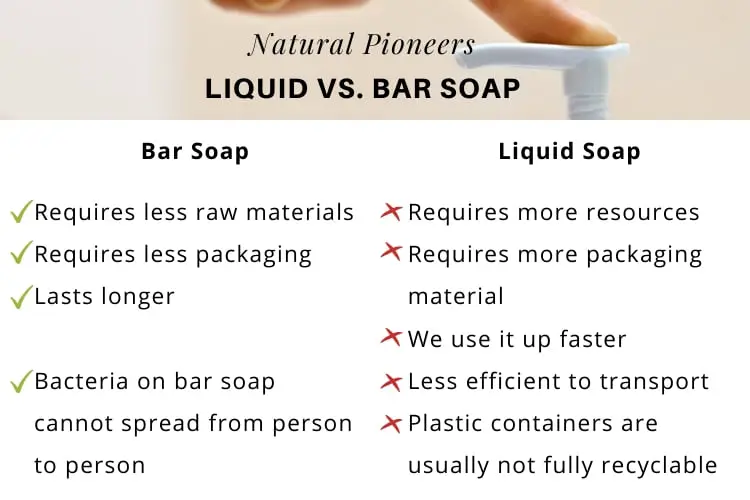
7. Bar Soap Is More Environmentally Friendly
According to a study conducted at the Swiss Federal Institute of Technology Zurich, bar soap production requires significantly less chemical raw materials. Moreover, bar soap is generally wrapped in eco-friendly paper whereas liquid soap comes in plastic bottles. Hence, bar soap requires far less packaging materials compared to liquid soap. [43]
Finally, when we consider the carbon footprint or impact on the environment, liquid soap is less efficient to transport and typically comes in a plastic container which is not completely recyclable. When it comes to the environment, bar soap is the clear winner!
>> Read more about: Can I use bar soap to wash my face?
8. Conclusion
The difference between liquid and bar soap lies in their ingredients. Ordinary soap is made by combining fats or oils and an alkali, such as lye. While bar soap is made with sodium hydroxide (lye), liquid soap is made with potassium hydroxide (another type of lye, also called potash or KOH).
Liquid soap and bar soap are equally effective. According to studies, soap and water reduce the presence of bacteria to 8%. Despite the rumors, bar soap does not transfer bacteria from person to person and bacteria cannot grow on a bar of soap. As far as antibacterial soap is concerned, studies show, that antibacterial soap is no more effective in reducing bacteria than regular soap.
Healthy skin has a pH between 4 and 5. To support our skin’s acid mantle, an ideal pH of a liquid or bar soap should be ≤ 5. The majority of soaps have a pH within the range of 9-10 and make our skin prone to damage and infection (acne, psoriasis and eczema.). Once damaged, it can take up to 14 hours to restore.
Natural bar soap contains most glycerin! Glycerin is a natural by-product when making soap and makes up 5-10% in soaps. Glycerin pulls water into the outer layer of your skin and traps it there. That means, it effectively hydrates and moisturizes our skin. Many popular soap brands extract glycerin for the use in cosmetics, resulting in extremely harsh, drying soaps. Many natural soap producers use a process called superfatting, resulting in about 1-20% extra nourishing “superfat”.
Conventional bar and liquid soaps sold in the US contain chemicals that are banned in other countries for their toxicity. Antibacterial agents, fragrances, surfactants, and preservatives are ingredients to avoid as they may cause hormone disruption, antibiotic resistance, reproductive disorders, disturbance in normal development of children, eye, nose, and throat irritation, kidney and liver damage, allergies, cancer, and more. Non-toxic soaps as featured in our resources are ideal.
From an environmental perspective, bar soap production requires significantly less chemical raw materials, and is packaged without the use of plastics as opposed to liquid soap bottles. Moreover, researchers found that when washing, we use 6x more liquid soap than bar soap. Liquid soap has a large carbon footprint as it is less efficient to transport and typically comes in a plastic container which is not completely recyclable. When it comes to the environment, bar soap is the clear winner!

